Objectives:
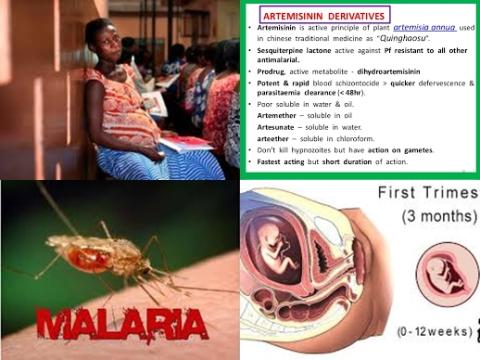
Artemisinin combination therapies (ACTs), the most efficacious antimalarials available, are the recommended first-line treatment for Plasmodium falciparum malaria except in the first trimester of pregnancy. Animal embryotoxicity data and the scarcity of safety data in human pregnancies, have prevented artemisinin derivatives from being recommended for malaria treatment in the first trimester except in lifesaving circumstances. Therefore, this review article (meta-analysis) has been conducted.
The aim of this review article is to compare the risk of miscarriage, stillbirth and major congenital anomaly (primary outcomes) among first-trimester pregnancies treated with artemisinin derivatives versus quinine or no antimalarial treatment.
Study design:
This review article included 5 prospective observational studies involving 30,618 pregnancies; 4 from sub-Saharan Africa (n = 6,666 pregnancies, 6 sites) and 1 from Thailand (n = 23,952).
Results and conclusions:
The investigators found no difference in the risk of miscarriage associated with the use of artemisinins anytime during the first trimester (n = 37/671) compared with quinine [adjusted hazard ratio = 0.73, 95% CI = 0.44 to 1.21, I2 = 0%, p = 0.228, n = 96/945].
The investigators found pregnancies treated with quinine during the first trimester were associated with significantly increased risk of 48% of miscarriage compared with pregnancies not treated with an antimalarial [adjusted hazard ratio = 1.48, 95% CI = 1.18 to 1.86]. However, in the sensitivity analysis the association between miscarriage and first-trimester quinine treatment compared with no antimalarial treatment was no longer significant when data from Thailand were omitted [adjusted hazard ratio = 2.12, 95% CI = 0.76 to 5.94, p = 0.153].
Significant because RR of 1 was not found in the 95% CI of 1.18 to 1.86. RR of 1 means no risk/association.
The investigators found pregnancies treated with artemisinins during the first trimester were not associated with an increased risk of miscarriage compared with pregnancies not treated with an antimalarial [adjusted hazard ratio = 1.16, 95% CI = 0.81 to 1.66].
Not associated because adjusted hazard ratio of 1 was found in the 95% CI of 0.81 to 1.66. Adjusted hazard ratio of 1 means no risk/association.
The investigators found no difference in the risk of stillbirth associated with the use of artemisinins anytime during the first trimester (n = 10/654) compared with quinine [adjusted hazard ratio = 0.29, 95% CI = 0.08 to 1.02, p = 0.053, n = 11/615].
The investigators found neither treatment with an artemisinin nor quinine was associated with an increased risk of stillbirths compared to pregnancies without any antimalarial treatment in the first trimester [adjusted hazard ratio = 0.65, 95% CI = 0.34 to 1.23 and adjusted hazard ratio = 1.35, 95% CI = 0.69 to 2.65, respectively].
The investigators found no difference in the risk of miscarriage and stillbirth combined (pregnancy loss) associated with the use of artemisinins anytime during the first trimester (n = 10/654) compared with quinine [adjusted hazard ratio = 0.58, 95% CI = 0.36 to 1.02, p = 0.099].
The investigators found the prevalence of major congenital anomalies was similar for first-trimester artemisinin [1.5%, 95% CI = 0.6% to 3.5%] and quinine exposures [1.2%, 95% CI = 0.6% to 2.4%].
The investigators concluded that first-trimester use of artemisinin derivatives is not associated with an increased risk of miscarriage or stillbirth compared to quinine. The data to date also indicate no difference in the prevalence of major anomalies between treatment groups in early pregnancy, although the numbers of major anomalies were small. Three-day artemisinin combination therapy (ACT) regimens are currently recommended to treat malaria in the second and third trimester. Expanding ACT recommendations to include the first trimester may outweigh the adverse outcomes of partially treated malaria due to poor adherence to 7 days oral quinine regimens in early pregnancy.
Original title:
First-trimester artemisinin derivatives and quinine treatments and the risk of adverse pregnancy outcomes in Africa and Asia: A meta-analysis of observational studies by Dellicour S, Sevene E, […], Stergachis A.
Link:
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5412992/
Additional information of El Mondo:
Find more information/studies on food fortification/malnutrition, pregnancy and study design/meta-analysis/significant right here.